Autoimmune Disease Series--
Myasthenia Gravis
Myasthenia gravis (MG) is a rare chronic autoimmune neuromuscular disorder caused by pathogenic immunoglobulin G antibodies attacking postsynaptic components at the neuromuscular junction (such as AChR and MuSK), leading to impaired neural signal transmission. Clinical manifestations include fluctuating muscle weakness and fatigue, commonly affecting eye muscles, bulbar muscles, and limb muscles. In severe cases, it can lead to respiratory failure. Approximately 85% of patients have generalized myasthenia gravis (gMG), with AChR antibody positivity being predominant, and those with MuSK antibody positivity exhibiting more severe symptoms. The pathogenesis of this disease involves antibody-mediated complement activation and the recycling and maintenance of pathogenic IgG by FcRn receptors, leading to sustained autoimmune damage.
Recent drug development progress in MG has focused on two core mechanisms: targeting the clearance of pathogenic antibodies and regulating the complement pathway, resulting in a treatment landscape dominated by FcRn inhibitors and complement C5 inhibitors:
● FcRn-targeted drugs accelerate the degradation of pathogenic antibodies by blocking FcRn-IgG binding, making them the most active area of current research and development. Representative drugs include efgartigimod, rozanolixizumab , and nipocalimab.
● Complement C5 inhibitors reduce the formation of membrane attack complexes by blocking the terminal complement cascade. Representative drugs include ravulizumab.
Future drugs for myasthenia gravis (MG) will evolve from broad-spectrum immunosuppression to precise targeting. FcRn and C5 inhibitors currently account for 67% of innovative drugs, while dual-target drugs and CAR-T therapies without chemotherapy are expected to further break through treatment bottlenecks for refractory patients.
Myasthenia Gravis Drug Targets in Development:
Myasthenia Gravis Drug Targets Related Products
● Target Proteins

Measured by its binding ability in a functional ELISA. Immobilized CD40 at 2 μg/ml can bind CD40L (CSB-MP004937HU3), the EC50 is 3.112-3.858 ng/ml.

Measured by its binding ability in a functional ELISA. Immobilized Human CLEC4C at 2μg/mL can bind Anti-CLEC4C recombinant antibody(CSB-RA855470MA1HU),the EC50 is 7.658-12.99 ng/mL.

Measured by its binding ability in a functional ELISA. Immobilized human CD20 at 2 μg/ml can bind Anti-CD20 recombinant antibody (CSB-RA015007A1HU), the EC50 is 3.243-7.085 ng/mL.
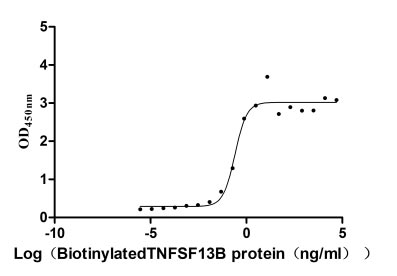
Measured by its binding ability in a functional ELISA. Immobilized human BCMA (CSB-MP023974HU1) at 5 μg/ml can bind Biotinylated human TNFSF13B, the EC50 is 0.1752-0.3657 ng/ml.