Autoimmune Disease Series--
Interstitial Lung Disease
Interstitial Lung Disease (ILD) comprises a heterogeneous group of disorders characterized by inflammation of the pulmonary parenchyma and progressive fibrosis. The core pathological mechanism involves repetitive alveolar epithelial injury followed by aberrant repair, leading to fibroblast activation, excessive deposition of extracellular matrix, and irreversible pulmonary scarring.
ILD encompasses over 200 subtypes, among which Idiopathic Pulmonary Fibrosis (IPF) is the most common and carries the poorest prognosis. It primarily affects individuals over 50 years of age, with a global prevalence of approximately 6.3 per 100,000 and a 5-year survival rate of only 20%-25%—lower than most cancers.
Clinical manifestations include progressive dyspnea, dry cough, and inspiratory crackles, with advanced stages potentially complicated by pulmonary hypertension and respiratory failure.
Recent drug development for ILD has focused on novel antifibrotic targets and disease-modifying therapies, with key advances including:
► PDE4B Inhibitors:
- Nerandomilast: Boehringer Ingelheim's oral selective PDE4B inhibitor, which elevates intracellular cAMP to suppress NF-κB/NFAT signaling and reduce pro-inflammatory cytokine release. Its Phase III FIBRONEER™-ILD trial met primary endpoints in February 2025, becoming the first successful Phase III novel drug in a decade. Applications for IPF and Progressive Pulmonary Fibrosis (PPF) indications have been submitted to regulatory authorities in China and the US.
- SYH2059: CSPC Pharmaceutical Group's highly selective PDE4B inhibitor with superior preclinical activity. Approved for clinical trials in China and the US in 2025, targeting ILD.
► Cell Therapy:
- NCR101 Injection: The world's first gene-edited induced pluripotent stem cell (iPSC)-derived mesenchymal-like cell (iMSC) therapy, engineered to enhance anti-inflammatory and tissue-repair functions. Received Chinese clinical approval in March 2025 for ILD treatment, overcoming limitations of traditional MSC heterogeneity and yield.
► Acute Exacerbation Management:
- Nintedanib: Systematic reviews demonstrate that initiating treatment during ILD acute exacerbation (AE-ILD) significantly reduces in-hospital mortality and shortens hospitalization potentially mediated through multi-kinase inhibition (PDGFR/VEGFR/FGFR) blocking fibrotic signaling.
► Emerging Targets:
- Anti-WISP1 Antibody (MTX-463): Developed by Lilly/Mediar, neutralizes WNT pathway protein WISP1 to inhibit fibroblast activation. Currently in Phase II trials.
- JAK Inhibitors (e.g., Ruxolitinib): Target JAK-STAT pathway to modulate macrophage polarization, showing 50% reduction in collagen deposition in preclinical models and exploring drug repurposing.
Interstitial Lung Disease Drug Targets in Development:
Interstitial Lung Disease Drug Targets Related Products
● Target Proteins

Measured by its binding ability in a functional ELISA. Immobilized CD147 at 2 μg/ml can bind Anti-CD147 recombinant Antibody, the EC50 is 21.95-33.12 ng/ml.

Measured by its binding ability in a functional ELISA. Immobilized Rhesus macaque CTGF at 1 μg/ml can bind Anti-CTGF recombinant antibody (CSB-RA006147MA2HU). The EC50 is 0.6886-0.7825 ng/mL.

Measured by its binding ability in a functional ELISA. Immobilized PD-L1 at 2 μg/ml can bind Anti- PD-L1 mouse monoclonal antibody(CSB-MA878942A1m,antigen from E.coli), the EC50 of human PD-L1 protein is 1.252-1.653 ng/mL.

The ED50 as determined by the dose-dependent stimulation of the proliferation of THP-1 cells is 2.880-4.115 ng/mL.

The ED50 as determined by the dose-dependent stimulation of the proliferation of TF-1 cells is 2.045-6.215 ng/mL

Measured by its binding ability in a functional ELISA. Immobilized human CD20 at 2 μg/ml can bind Anti-CD20 recombinant antibody (CSB-RA015007A1HU), the EC50 is 3.243-7.085 ng/mL.

Measured by its binding ability in a functional ELISA. Immobilized CD73 at 2 μg/ml can bind Anti- CD73 Rabbit Monoclonal Antibody (CSB-RA978310A0HU), the EC50 is 3.212-4.525 ng/ml.
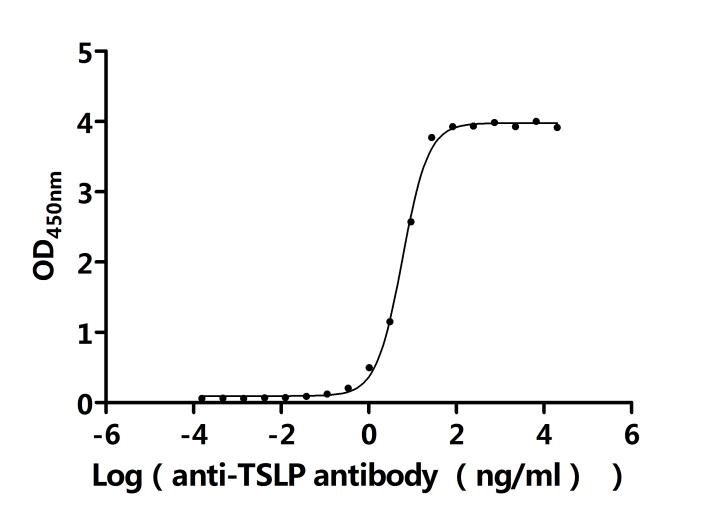
Measured by its binding ability in a functional ELISA.Immobilized Human TSLP at 2 μg/ml can bind Anti-TSLP recombinant antibody(CSB-RA025141MA2HU). The EC50 is 5.487-6.214 ng/mL.