| Method |
Technical Features |
Applications |
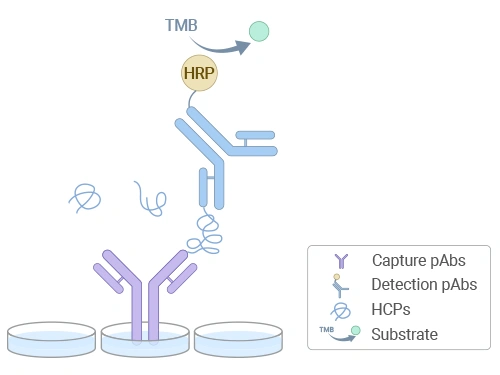
| ELISA (Enzyme-Linked Immunosorbent Assay) |
- Based on a double-antibody sandwich method, high sensitivity (detection and quantification limits as low as ng/mL);
- Simple operation, standardized kits available;
- Relies on high-quality antibodies and specificity, requiring antibody coverage validation.
|
- Quality control, process optimization, final product release testing;
- Recommended by regulatory guidelines (e.g., USP, ICH).
|
| Mass Spectrometry (LC-MS/MS) |
- High resolution, capable of identifying specific HCP types, antibody-independent, comprehensive coverage;
- Requires specialized equipment and data processing support.
|
- Process development for HCP characterization, high-risk HCP analysis (e.g., PLBL2), orthogonal validation of ELISA results.
|
| Western Blot |
- Visualized results, detects specific HCPs, provides molecular weight information;
- Lower sensitivity (requires ng-level protein);
- Limited to known HCPs.
|
- Screening specific HCPs, validating ELISA results, monitoring HCP in process intermediates.
|
| Two-Dimensional Gel Electrophoresis (2D-PAGE) |
- Combines isoelectric focusing and SDS-PAGE to separate HCPs and generate 2D maps;
- Cannot quantify directly, requires mass spectrometry for identification.
|
- HCP characterization during process development, preliminary analysis of complex samples.
|
| Capillary Electrophoresis (CE) |
- Fast, requires minimal sample volume (nL level), highly automated;
- Lower sensitivity (requires μg-level protein), often used in combination with other methods.
|
- Rapid HCP screening, small-scale sample analysis, quantification with mass spectrometry.
|
| HPLC |
- Useful for evaluating HCP removal efficiency, standardized operation;
- Cannot distinguish HCP types, depends on elution conditions.
|
- Monitoring purity of process intermediates, optimizing HCP removal processes.
|