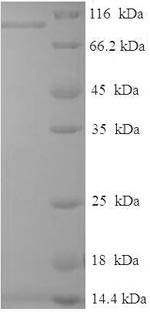
The production of the recombinant human RAF proto-oncogene serine/threonine-protein kinase (RAF1) begins with obtaining the target gene, which encodes the 1-648aa of the human RAF1. This target gene along with an N-terminal 6xHis-SUMO-tag gene is cloned into an expression vector, which is then introduced into E. coli cells through transformation. Once inside the host cell, the gene is expressed, and the protein of interest is produced. The generated protein is harvested from the cell culture and purified, often using affinity chromatography. Finally, the recombinant RAF1 protein's purity is determined by SDS-PAGE, reaching up to 90%.
RAF1 acts as a signaling hub in the MAPK/ERK pathway, linking the GTPase RAS to the extracellular signal-regulated kinase (ERK) module, which includes kinases of the RAF family, MEK, and ERK [1]. Activation of RAF1 by GTP-bound RAS leads to the phosphorylation of serine residues on MEK1 and MEK2, further propagating the signaling cascade [2]. RAF1 is involved in various cellular processes such as proliferation, differentiation, apoptosis, motility, and metabolism [3].
Mutations in RAF1 have been associated with different diseases, including Noonan syndrome and hypertrophic cardiomyopathy [4][5][6]. RAF1 amplification has been linked to driving a subset of bladder tumors, making it a potential target for MAPK-directed therapeutics [7]. Evidence has shown that RAF1 is implicated in colorectal cancer, where its inhibition can restore apicobasal polarity and impair tumor growth [8].
RAF1's function extends beyond its kinase activity, as it has been shown to have kinase-independent functions mediated through protein-protein interactions [9]. RAF1 dimerization, especially with BRAF, is crucial in the pathogenesis of Noonan syndrome, emphasizing the importance of RAF1-BRAF interactions in disease mechanisms [10].
References:
[1] A. Varga, K. Ehrenreiter, B. Aschenbrenner, P. Kocieniewski, M. Kochańczyk, T. Lipniackiet al., Raf1/braf dimerization integrates the signal from ras to erk and rokα, Science Signaling, vol. 10, no. 469, 2017. https://doi.org/10.1126/scisignal.aai8482
[2] H. Yagasaki, T. Nakane, Y. Hasebe, A. Watanabe, H. Kise, T. Todaet al., Co‐occurrence of hypertrophic cardiomyopathy and myeloproliferative disorder in a neonate with noonan syndrome carrying thr73ile mutation in ptpn11, American Journal of Medical Genetics Part A, vol. 167, no. 12, p. 3144-3147, 2015. https://doi.org/10.1002/ajmg.a.37295
[3] Y. Huang, X. Guo, B. Han, X. Zhang, S. An, X. Zhanget al., Decoding the full picture of raf1 function based on its interacting proteins, Oncotarget, vol. 8, no. 40, p. 68329-68337, 2017. https://doi.org/10.18632/oncotarget.19353
[4] R. Hopper, J. Feinstein, M. Manning, W. Benitz, & L. Hudgins, Neonatal pulmonary arterial hypertension and noonan syndrome: two fatal cases with a specific raf1 mutation, American Journal of Medical Genetics Part A, vol. 167, no. 4, p. 882-885, 2015. https://doi.org/10.1002/ajmg.a.37024
[5] H. Jaouadi, A. Chehida, L. Kraoua, H. Etchevers, L. Argiro, N. Kasdallahet al., A severe clinical phenotype of noonan syndrome with neonatal hypertrophic cardiomyopathy in the second case worldwide with raf1 s259y neomutation, Genetics Research, vol. 101, 2019. https://doi.org/10.1017/s0016672319000041
[6] F. Harms, M. Alawi, D. Amor, T. Tan, G. Čuturilo, C. Lißewskiet al., The novel raf1 mutation p.(gly361ala) located outside the kinase domain of the cr3 region in two patients with noonan syndrome, including one with a rare brain tumor, American Journal of Medical Genetics Part A, vol. 176, no. 2, p. 470-476, 2017. https://doi.org/10.1002/ajmg.a.38569
[7] R. Bekele, A. Samant, A. Nassar, J. So, E. Garcia, C. Curranet al., Raf1 amplification drives a subset of bladder tumors and confers sensitivity to mapk-directed therapeutics, Journal of Clinical Investigation, vol. 131, no. 22, 2021. https://doi.org/10.1172/jci147849
[8] T. Borovski, T. Vellinga, J. Laoukili, E. Santo, S. Fátrai, S. Schelvenet al., Inhibition of raf1 kinase activity restores apicobasal polarity and impairs tumour growth in human colorectal cancer, Gut, vol. 66, no. 6, p. 1106-1115, 2016. https://doi.org/10.1136/gutjnl-2016-311547
[9] L. Iglesias-Martinez, N. Rauch, K. Wynne, B. McCann, W. Kölch, & J. Rauch, Interactome dynamics of raf1-braf kinase monomers and dimers, Scientific Data, vol. 10, no. 1, 2023. https://doi.org/10.1038/s41597-023-02115-0
[10] X. Wu, J. Yin, J. Simpson, K. Kim, S. Gu, J. Honget al., Increased braf heterodimerization is the common pathogenic mechanism for noonan syndrome-associated raf1 mutants, Molecular and Cellular Biology, vol. 32, no. 19, p. 3872-3890, 2012. https://doi.org/10.1128/mcb.00751-12