Tumor Necrosis Factor Receptor 1 (TNFR1) is a key molecule mediating apoptosis, necrosis, and inflammatory responses. It plays a complex role in pathological processes such as cancer, autoimmune diseases, and infections. In tumors, it can induce apoptosis through its death domain while also activating the NF-κB pathway to promote survival. This "dual role" makes it a highly promising therapeutic target. Additionally, TNFR1-mediated cytokine storms are considered a key mechanism of tissue damage in severe infections like COVID-19, making inhibitors targeting it a former research hotspot.
In recent years, TNFR1-targeting strategies have shifted from broad TNF inhibition towards more precise interventions. The research frontier focuses on developing drugs that specifically block TNFR1 signaling without affecting the protective functions of TNFR2, as well as utilizing new technologies like cellular engineering to intelligently "reprogram" inflammatory signaling. These advances are propelling the field into a new stage of "precision regulation" and "pathological signal transformation."
Against this backdrop, CUSABIO is committed to providing high-quality TNFR1-related proteins and tools to support cutting-edge mechanistic research and drug development targeting this molecule. Our products are designed to help researchers analyze the TNFR1 pathway more deeply and precisely, laying the groundwork for the discovery of innovative therapies.
This article will review the structure and function of TNFR1, the signaling pathways it mediates, its "dual role" in diseases such as cancer and COVID-19, and discuss therapeutic strategies and the latest advances in targeting TNFR1.
1. TNFR1 Structure and Function
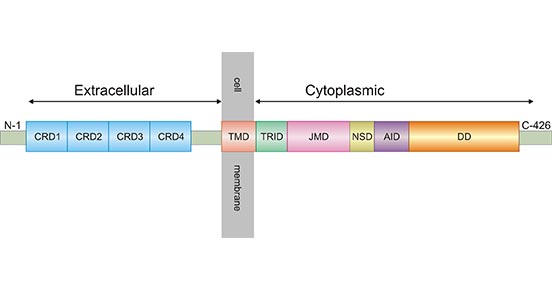
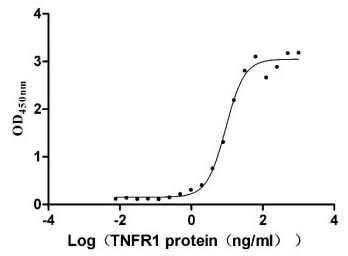
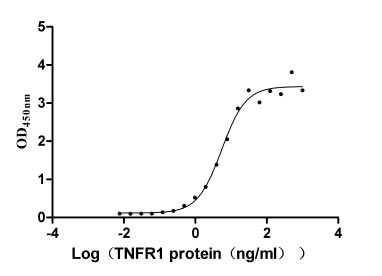
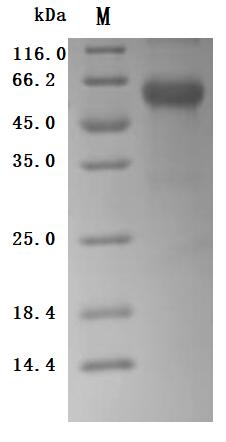
TNFR1 (as known as TNFRSF1A, CD120a or p55), a member of the tumor necrosis factor (TNF) receptor superfamily, is a type I transmembrane glycoprotein with a molecular weight of 55 ku [2]. TNFR1 has extracellular, transmembrane and intracellular domains. The extracellular region is approximately 182 amino acids long and consists of four cysteine rich domains (CRD1-CRD2-CRD3-CRD4) starting from the N-terminus. The transmembrane region lies between the extracellular and intracellular domains and it is approximately 22 amino acids long (residues 183-205). The intracellular/cytoplasmic domain of TNFR1 is about 223 amino acids long and is important for activation of several downstream signaling pathways via its conserved death domain (DD). In resting cells, the silencer of death domain (SODD) binds and inhibits the function of death domain (Figure 1) [3, 4].
Figure 1. Schematic diagram for TNFR1 structure
*The figure is derived from ResearchGate publication [4]
TNFR1 is widely distributed on the surfaces of various immune system-related cell types such as macrophages and regulatory T cells as well as in many tumor cells [5]. TNFR1 mainly mediates apoptotic signaling pathway, leading to apoptosis, which plays an important role in anti-tumor and anti-viral. It is also involved in autoimmune diseases, being a key factor in the induction of rheumatoid arthritis (RA) [6] and systemic lupus erythematosus (SLE) [7]. TNFR1, a double-edged sword, its underlying molecular mechanism still remains unclear.
2. The Ligand of TNFR1
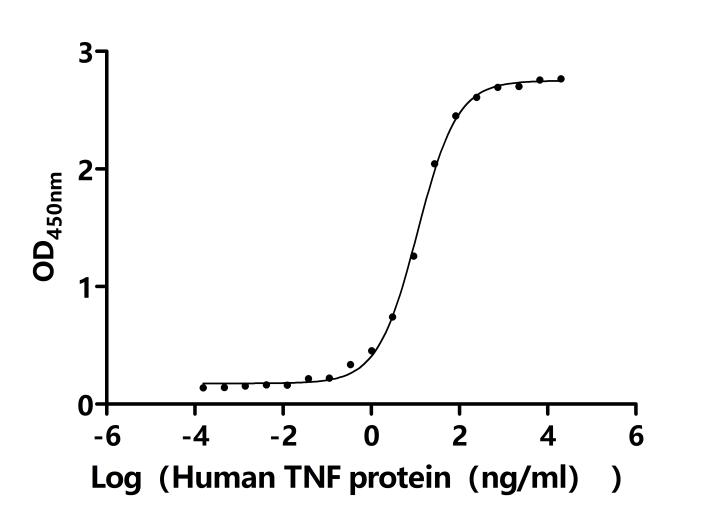
TNF is a compact trimer composed of three identical subunits [8]. TNF contains TNF-α and TNF-β, which are type II membrane proteins. TNF-α is mostly produced by activated macrophages, whereas TNF-β is produced by activated lymphocytes [9] Currently, TNF is often called TNF-α since the TNF-β level has been shown to be quite less than TNF-α [10].
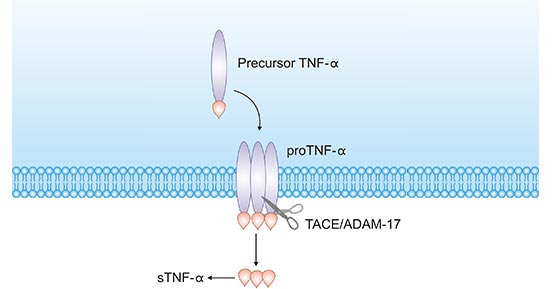
TNFα exists in two forms, transmembrane TNF-α (tmTNF-α) and soluble TNF-α (sTNF-α). tmTNF-α is the precursor form of sTNF-α. After processing by TACE-converting enzyme (also known as ADAM17/CD156q), sTNF-α is generated (Figure 2) [11].
Figure 2. tmTNF-α to sTNF-α process
*The figure is derived from the Ochsner Journal publication [11]
Both types of TNFα; interact with receptors (TNFR1 or TNFR2) to induce biological effects, but since TNFR2 lacks a death domain, TNFR1 becomes the primary receptor mediating TNF-α; activity [12, 13, 14]. Binding of TNFR1 to TNF-α; causes changes in the conformation of the intracellular region, which attributes to the TNFR1 activation [15].
3. TNFR1-Mediated Signaling Pathways
3.1 TNFR1-sTNF-α Signaling Pathway
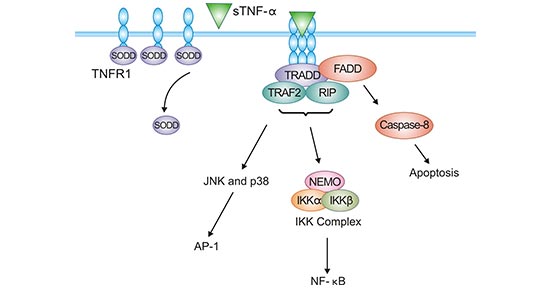
Regarding the TNFR1-sTNF-α signaling pathway, it has been well-defined in recent years. Without sTNF-α stimulation, the DD of TNFR1 is occupied by the silencer of the death domain (SODD), which blocks the TNFR-related death domain protein (TRADD) from combing with TNFR1. TNFR1 signaling pathway is restrained [16]. Upon sTNF-α stimulation, SODD dissociates from the TNFR1 DD. TNFR1 recruits TRADD and the TNFR1 signaling pathway is activated (Figure 3) [17].
Once TNFR1/sTNF-α is activated, TRADD can recruit receptor-associated protein-1 (RIP1) and receptor-associated factor 2 (TRAF2), which activates NF-κB. The activation of NF-κB promotes target gene transcription, which resists apoptosis and promotes survival (Figure 3) [4, 17]
In addition, TNFR1 binds to DD of FADD (Fas-associated death domain protein) through DD of TRADD. FADD can bind to DED of caspase-8 through its death-effective DED domain, which activates caspase-8, leading to apoptosis or programmed necrosis (necroptosis) (Figure 3) [4, 17].
Figure 3. TNFR1-sTNF-α signaling pathway
*The figure is derived from Cytokine publication [17]
3.2 TNFR1-tmTNF-α Signaling Pathway
Regarding the signaling pathway of TNFR1-tmTNF-α, the current mechanism is unclear. Some studies have found that the TNFR1-tmTNF-α signaling pathway is different from TNFR1-sTNF-α, which can mediate two distinct signaling pathways, survival or apoptosis. While TNFR1-tmTNF-α only mediates apoptosis and does not activate NF-κB. Although TNFR1 binding to both types of TNF can cause apoptosis, they have different signaling mechanisms and different cellular localization [4].
4. The Role of TNFR1 in Tumors
In recent years, more and more researchers have found that TNFR1 is widely involved in the pathophysiological processes of various diseases, especially in tumors. Thus its regulatory role has attracted attention. Extensive data have shown that TNFR1 can determine different cell fates in different cancer cells. Although TNFR1 contains the death domain, TNFR1 can also exert pro-inflammatory and oncogenic effects through different signaling pathways. Therefore, TNFR1 does not always exert pro-apoptotic biological effects in cancer.
In malignant astrocytomas, TNFR1 binds to TNF, activates the NF-κB pathway, and inhibits apoptosis of tumor cells. The study suggests that TNFR1 may be involved in the formation of low-grade malignant astrocytomas [18]. Another study found that in gastric cancer cells, the expression level of TNFR1 was correlated with the degree of differentiation of gastric cancer cells [19]; in colorectal cancer, patients with high TNFR1 expression had a higher survival rate [20]. A recent study found that in renal clear cell carcinoma, TNFR1 promotes tumor-like lesions and induces drug resistance [21]. In a mouse model experiment, researchers evaluated the role of TNFR1 in hepatocellular carcinoma. The data showed that TNFR1 deficiency significantly reduced tumorigenesis in mice, suggesting that TNFR1-mediated signaling pathways promote hepatocarcinogenesis [22].
TNFR1 is involved in an extremely complex biological process, and its functions is often influenced by many uncontrollable factors (e.g., individualized patient differences). The specified roles of TNFR1 in human tumors are not well understood. Whether it mediates cell proliferation or apoptosis in different tumors requires more studies to further elucidate. Therefore, the application of TNFR1 antibodies or anti-TNFR1 antibodies is of great significance in tumor therapy.
5. Conclusion
TNFR1, as a core molecule regulating cell death and inflammatory responses, holds significant therapeutic value in cancer, autoimmune diseases, and infectious diseases due to its complex "dual role." As research progresses, future developments in this field will focus on precision, intelligence, and individualization.
Future research strategies will move beyond traditional broad-spectrum inhibition toward spatiotemporally precise interventions targeting the TNFR1 signaling pathway. For instance, developing drugs that selectively block the binding of specific TNF forms (such as soluble TNF) to TNFR1 could help suppress pathogenic inflammation while preserving its physiological regulatory and tissue repair functions. Meanwhile, cell therapies represented by CAR-M have pioneered a new paradigm of "sensing and reprogramming" pathological signals, representing an intelligent direction in inflammatory regulation.
Furthermore, fundamental research on specific signaling complexes of TNFR1 in different disease microenvironments will be deeply integrated with clinical translation efforts, such as biomarker exploration. This integration is key to developing more effective drugs, enabling patient stratification, and overcoming heterogeneity in treatment responses. The therapeutic applications of TNFR1 are also expected to expand beyond current areas to include neurodegenerative diseases, fibrosis, and other indications.
CUSABIO will continue to provide high-quality research tools and protein products for this promising target, supporting end-to-end innovation from mechanism exploration to therapeutic development, and facilitating the early translation of next-generation TNFR1-targeted strategies for patient benefit.
References
[1] Ding, Husheng, et al. "CDK2-mediated upregulation of TNFa as a mechanism of selective cytotoxicity in acute leukemia." Cancer Research. 2021.
[2] Speeckaert, Marijn M., et al. "Tumor necrosis factor receptors: biology and therapeutic potential in kidney diseases." American journal of nephrology 36.3 (2012): 261-270.
[3] Gray, Patrick W., et al. "Cloning of human tumor necrosis factor (TNF) receptor cDNA and expression of recombinant soluble TNF-binding protein." Proceedings of the National Academy of Sciences 87.19 (1990): 7380-7384.
[4] Negm, Ola Hamdy El-Shahat. "Investigations of Signalling Pathways Activation by Mutant Tumour Necrosis Factor Receptors." (2011).
[5] Sedger, Lisa M., and Michael F. McDermott. "TNF and TNF-receptors: From mediators of cell death and inflammation to therapeutic giants–past, present and future." Cytokine & growth factor reviews 25.4 (2014): 453-472.
[6] Fischer, Roman, Roland E. Kontermann, and Olaf Maier. "Targeting sTNF/TNFR1 signaling as a new therapeutic strategy." Antibodies 4.1 (2015): 48-70.
[7] Deng, Guo-Min, et al. "Lupus serum IgG induces skin inflammation through the TNFR1 signaling pathway." The Journal of Immunology 184.12 (2010): 7154-7161.
[8] Jones, E. Y., D. I. Stuart, and NPC WALKER. "The structure of tumour necrosis factor-implications for biological function." Journal of Cell Science 1990.Supplement 13 (1990): 11-18.
[9] Li, Kang, et al. "The involvement of TNF-α and TNF-β as proinflammatory cytokines in lymphocyte-mediated adaptive immunity of Nile tilapia by initiating apoptosis." Developmental & Comparative Immunology 115 (2020): 103884.
[10] Wang, Xia, and Yong Lin. "Tumor necrosis factor and cancer, buddies or foes? 1." Acta Pharmacologica Sinica 29.11 (2008): 1275-1288.
[11] Shuh, Maureen, et al. "Tumor necrosis factor-α: life and death of hepatocytes during liver ischemia/reperfusion injury." Ochsner Journal 13.1 (2013): 119-130.
[12] Naudé, Petrus JW, et al. "Tumor necrosis factor receptor cross‐talk." The FEBS journal 278.6 (2011): 888-898.
[13] Gane, Jennie M., Robert A. Stockley, and Elizabeth Sapey. "TNF-α autocrine feedback loops in human monocytes: the pro-and anti-inflammatory roles of the TNF-α receptors support the concept of selective TNFR1 blockade in vivo." Journal of immunology research 2016 (2016).
[14] Van Hauwermeiren, Filip, Roosmarijn E. Vandenbroucke, and Claude Libert. "Treatment of TNF mediated diseases by selective inhibition of soluble TNF or TNFR1." Cytokine & growth factor reviews 22.5-6 (2011): 311-319.
[15] MacEwan, David J. "TNF ligands and receptors-a matter of life and death." British journal of pharmacology 135.4 (2002): 855-875.
[16] Miki, Kiyoshi, and Edward M. Eddy. "Tumor necrosis factor receptor 1 is an ATPase regulated by silencer of death domain."
Molecular and cellular biology 22.8 (2002): 2536-2543.
[17] Li, Hongxiu, and Xin Lin. "Positive and negative signaling components involved in TNFα-induced NF-κB activation." Cytokine 41.1 (2008): 1-8.
[18] Yang, Zijun, et al. "Phosphorylated form of pyruvate dehydrogenase α1 mediates tumor necrosis factor α‑induced glioma cell migration." Oncology Letters 21.3: 1-1.
[19] Teng, Chih-Chuan, et al. "Novel regulator role of CIL-102 in the epigenetic modification of TNFR1/TRAIL to induce cell apoptosis in human gastric cancer." Food and Chemical Toxicology 147 (2020): 111856.
[20] Yun, Hyung-Mun, et al. "IL-32α suppresses colorectal cancer development via TNFR1-mediated death signaling." Oncotarget 6.11 (2015): 9061.
[21] Hwang, Hee Sang, et al. "Involvement of the TNF-α Pathway in TKI Resistance and Suggestion of TNFR1 as a Predictive Biomarker for TKI Responsiveness in Clear Cell Renal Cell Carcinoma." Journal of Korean medical science 35.5 (2020).
[22] Bluemel, Sena, et al. "Tumor necrosis factor alpha receptor 1 deficiency in hepatocytes does not protect from non-alcoholic steatohepatitis, but attenuates insulin resistance in mice." World Journal of Gastroenterology 26.33 (2020): 4933.



Comments
Leave a Comment